|
Also used in street lights, batteries, table salt (NaCl), and glass. Liquid sodium is sometimes used to cool nuclear reactors. Uses of Sodium: Used in medicine, agriculture and photography.Primary mining areas are Germany, Poland, Kenya USA. Approximate annual world wide production: sodium metal, 200,000 tons salt, 168,000,000 tons sodium carbonate, 29,000,000 tons. Sources of Sodium: Obtained by electrolysis of melted sodium chloride (salt), borax and cryolite.Name Origin: From soda (Na2CO3) Na from Latin natrium.Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Vapor Pressure = C Regulatory / Health.Enthalpy of Vaporization: 89.04 kJ/mole.Enthalpy of Atomization: 108.4 kJ/mole 25☌.Description: Soft silvery white alkali metal that reacts violently with water and oxidises rapidly when cut.Conductivity Electrical: 0.21 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 70.6E -6.Valence Electron Potential (-eV): 14.1 Physical Properties of Sodium.Electronegativity: 0.93 (Pauling) 1.01 (Allrod Rochow).Electrochemical Equivalent: 0.85775g/amp-hr.Valence Electrons: 3s 1 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 12.Number of Electrons (with no charge): 11.Electrons per Energy Level: 2,8,1 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 1.Crystal Structure: Cubic body centered.Cross Section (Thermal Neutron Capture) σ a/ barns : 0.53.Swedish: Natrium Atomic Structure of Sodium.Series: Alkali Metals Sodium's Name in Other Languages.Common Chemical Compounds of Sodium Overview of Sodium.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Skip to site menu on this page Periodic Table of Elements Element Sodium - NaĬomprehensive data on the chemical element Sodium is provided on this page including scores of properties, element names in many languages, most known nuclides of Sodium.The barium cation is written Ba 2+, not Ba +2. Note the convention of first writing the number and then the sign on a multiply charged ion. Figure 3.3 “Predicting Ionic Charges” shows how the charge on many ions can be predicted by the location of an element on the periodic table. On the other side of the periodic table, the next-to-last column, the halogens, form ions having a 1− charge. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. 
For example, all ions made from alkali metals, the first column on the periodic table, have a 1+ charge. Thus, the periodic table becomes a tool for remembering the charges on many ions. 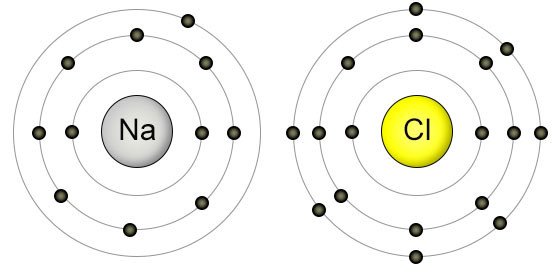
In many cases, elements that belong to the same group (vertical column) on the periodic table form ions with the same charge because they have the same number of valence electrons. In macroscopic samples of sodium chloride, there are billions and billions of sodium and chloride ions, although there is always the same number of cations and anions. The number of electrons lost by the sodium atom (one) equals the number of electrons gained by the chlorine atom (one), so the compound is electrically neutral. Notice that there are no leftover electrons. 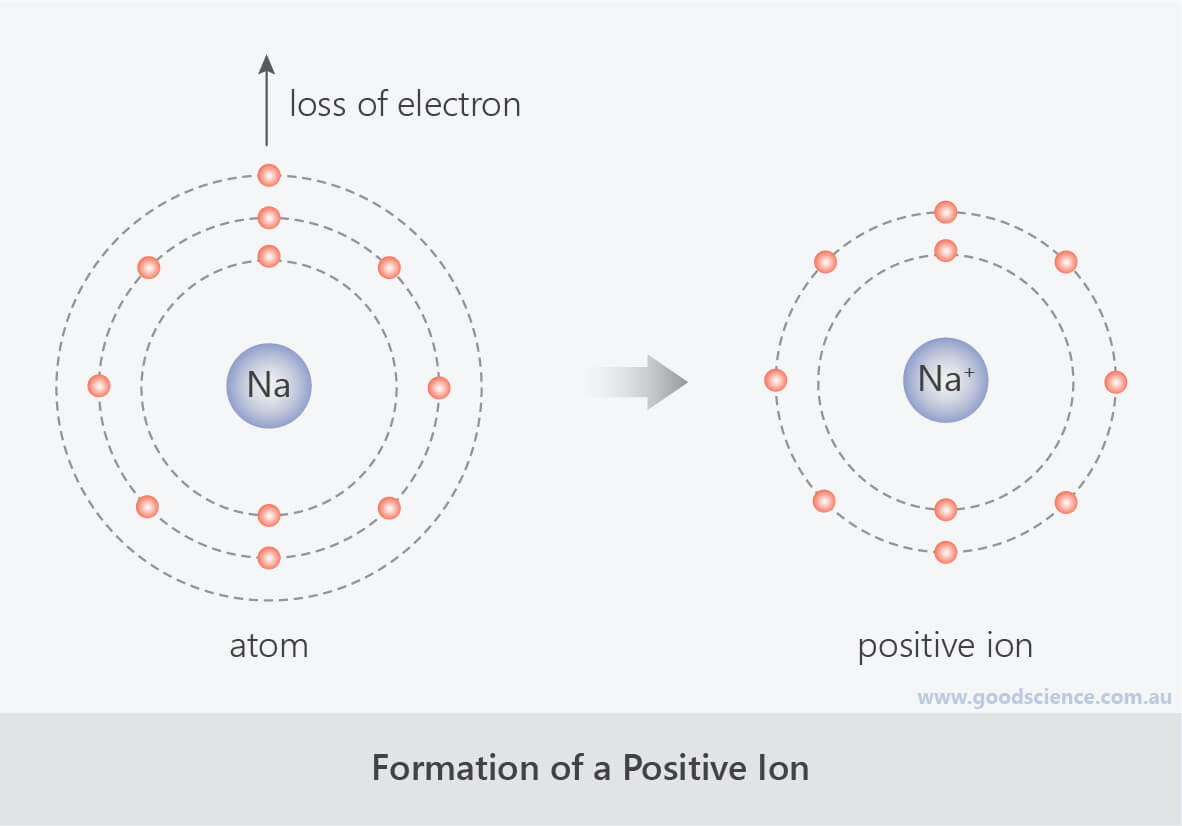
The resulting combination is the compound sodium chloride. With two oppositely charged ions, there is an electrostatic attraction between them because opposite charges attract. On the right, the chloride ion has 18 electrons and has a 1− charge. Most nonmetals become anions when they make ionic compounds.įigure 3.2 The Formation of a Chlorine Ion. On the left, the chlorine atom has 17 electrons. Negatively charged ions are called anions. When these atoms gain electrons, they acquire a negative charge because they now possess more electrons than protons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they have an octet. Most metals become cations when they make ionic compounds. Positively charged ions are called cations. Atoms that lose electrons acquire a positive charge as a result because they are left with fewer negatively charged electrons to balance the positive charges of the protons in the nucleus. In cases where an atom has three or fewer valence electrons, the atom may lose those valence electrons quite easily until what remains is a lower shell that contains an octet. Some atoms have only a few electrons in their outer shell, while some atoms lack only one or two electrons to have an octet. Most atoms do not have eight electrons in their valence electron shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
